|
10/30/2022 0 Comments Cracking of dodecane to make ethene 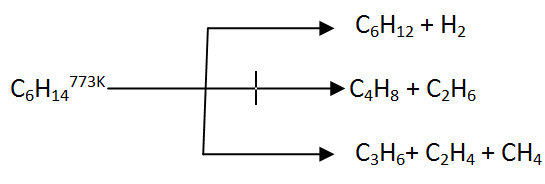
In both cases, the Langmuir-Hinshelwood-Hougen-Watson (LH) rate expressions are applied for the kinetic description of the system 36, 37. The second model is based on the modified network, including cracking reactions. The first model is based on the reaction network suggested by Iwamoto, including metathesis. The focus of this work will be the analysis and parametrization of two detailed kinetic models. So far, no reliable kinetic model equations were provided for predicting the direct conversion of ethene on Ni/AlMCM-41 (Si/Al = 60) to propene. These cycles provide a suitable basis for the derivation of similar kinetic rate laws. Both catalytic cycles are shown and discussed in Fig. In comparison to the cracking mechanism described, the metathesis is thoroughly investigated 33- 35. The cracking mechanism is considered to be based on the protolytic scission of the double-bonded atoms, resulting in separate olefinic molecules 31, 32. 
Several investigations exploiting MCM 26, 27 and ZSM 28- 30 have been performed. The applied aluminized MCM-41 belongs to a class of supports catalyzing this type of reaction 25. Recently, it was assumed that catalytic cracking of long-chain olefins to the desired short-chain olefins occurs 24. Buluchevski 23 applied PdO-Re 2O 7-B 2O 3-Al 2O 3 for the liquid phase reaction of ethene to propene. The metathesis reaction as one of the most important organic reactions of the last century was kinetically investigated by, e.g., Lwin 21 and Kapteijn 22. 20, is the application of alkanes, like hexane and heptane. Therefore, van Speybroeck 19 derived principle kinetics for applied zeolites. The methanol-to-olefin process is an exciting alternative to the well-known feedstock. Another compelling catalyst is SAPO-34 that was used by Mousavi 17 for the methanol-to-olefin process and by Zhou 18 for the reaction of light alkenes. The ZSM-5 was also applied by Beirnaert 13 and Aretin 14 for the cracking of hexene, Epelde 15 for the transformation of 1-butene and Gayubo 16 for ethanol conversion to olefins. Perez-Uriarte 12 used HZSM-5 for the conversion of dimethyl ether (DME) to olefins. Many research groups investigated the transformation of hydrocarbons to light olefins for various reaction types. Therefore, a more detailed reaction network is desirable, considering the relevant side reactions and the ongoing mechanism as the focus of this present contribution. Additionally, it neglects that nickel is not a typical active metal for metathesis reactions. The relatively simple network postulated does not consider the formation of essential by-products and their influence on the course of the reactions. This catalytic cycle has been discussed in more detail in, e.g., 2, 10, 11. It is supposed that nickel initiates the metathesis step. Finally, a metathesis step of the generated 2-butene and unconverted ethene results in propene. Initially, ethene is dimerized to 1-butene by the nickel ion of the catalyst, followed by the isomerization of 1-butene to cis-/ trans-butene catalyzed by the acidic sites. 1).Ĭatalytic reaction networks based on dimerization, isomerization combined with metathesis for network I, proposed by Iwamoto, and cracking for the modified network II, proposed in a previous contribution 24. 3 consists of dimerization, isomerization and metathesis reaction steps (see Fig. The widely accepted mechanism proposed by Iwamoto et al. To design and optimize the reaction system, a quantitative description of the underlying reaction mechanisms and kinetics is necessary. 2 studied the characteristics of Ni/MCM-41 and its aluminized counterpart Ni/AlMCM-41. 41) and found it to be active for the ETP reaction. 3, 7 investigated nickel-doped MCM-41 (Mobil Composition of Matter No. 6 presented silica aluminophosphate microporous molecular sieves, e.g., SAPO-34 (small-pore molecular sieves-34) and H-ZSM-5 (Zeolite Socony Mobil-5) zeolites, as highly active catalysts for the ETP reaction, but also faced severe deactivation with time on stream. have reported the direct transformation of ethene into propene over a tungsten hydride supported on alumina 4. 
Significant research was dedicated to developing suitable catalytic systems. The direct conversion of ethene to propene (ETP) 2, 3 is an attractive possibility, exploiting the availability of ethene. Therefore, new efficient on-demand technologies are required. The steadily growing demand cannot be easily met by the current production technologies 1, like steam cracking. It is mainly used for the production of the highly demanded polypropylene and propene oxide. Propene has become one of the central building blocks for the petrochemical industry 1. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
